For the H 2 molecule, the distance is 74 pm (picometers, 10 -12 meters). 
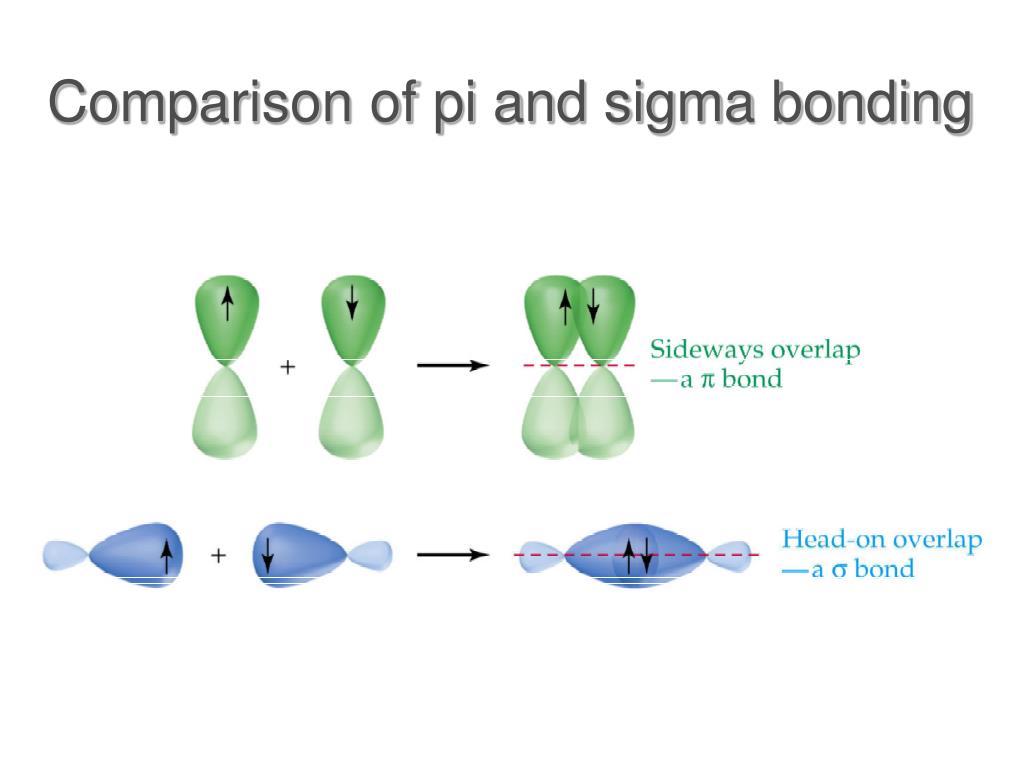
This optimal internuclear distance is the bond length. There is a defined optimal distance between the nuclei in which the potential energy is at a minimum, meaning that the combined attractive and repulsive forces add up to the greatest overall attractive force. When the two nuclei are ‘too close’, we have an unstable, high-energy situation. This lowers the potential energy of the system, as new, attractive positive-negative electrostatic interactions become possible between the nucleus of one atom and the electron of the second.īut something else is happening at the same time: as the atoms get closer, the repulsive positive-positive interaction between the two nuclei also begins to increase.Īt first this repulsion is more than offset by the attraction between nuclei and electrons, but at a certain point, as the nuclei get even closer, the repulsive forces begin to overcome the attractive forces, and the potential energy of the system rises quickly. As they move closer and closer together, orbital overlap begins to occur, and a bond begins to form. How far apart are the two nuclei? If they are too far apart, their respective 1 s orbitals cannot overlap, and thus no covalent bond can form - they are still just two separate hydrogen atoms. These two electrons are now attracted to the positive charge of both of the hydrogen nuclei, with the result that they serve as a sort of ‘chemical glue’ holding the two nuclei together. When we say that the two hydrogen nuclei share their electrons to form a covalent bond, what we mean in valence bond theory terms is that the two spherical 1 s orbitals (the grey spheres in the figure below) overlap, and contain two electrons with opposite spin. The simplest case to consider is the hydrogen molecule, H 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
